FDA Inspection Strategy and FDA Consulting
FDA Inspection Playbook
An FDA inspection is not scheduled around your readiness—it is triggered by risk, history, and regulatory priorities. While federal law references a two-year inspection cycle for drug manufacturers, FDA does not follow a predictable calendar, and medical device inspections are entirely risk-based. This means your next inspection can happen when you least expect it.
An FDA inspection playbook is not a checklist—it is a disciplined approach to operating under constant inspection conditions. It defines how your systems perform, how your records support decisions, and how your team responds when challenged.
Organizations that rely on timing or past inspection gaps are taking unnecessary risk. FDA consulting provides clarity before that moment arrives—aligning your operations with how FDA actually evaluates compliance and ensuring your organization is prepared to perform, not react.
Who Is FDA Office of Inspections and Investigations (OII)
The FDA Office of Inspections and Investigations (OII), formerly part of the Office of Regulatory Affairs (ORA), is the frontline of FDA enforcement. This is the group that shows up at your facility, conducts inspections, collects evidence, and determines whether your operations meet regulatory requirements. OII operates globally, covering food, drugs, medical devices, and other regulated products. They do not just inspect—they investigate, screen imports, collaborate with other agencies, and respond to public health risks. Every 33 minutes, OII initiates an inspection or regulatory activity, making it a constant and active presence across industries.
Understanding OII is critical. FDA is not theoretical—this is the operational arm that evaluates your systems, your data, and your decisions in real time.
OII Conducts Inspections That Drive Regulatory Action
OII investigators conduct inspections of medical device and pharmaceutical manufacturers worldwide. They assess quality systems, records, and processes against FDA requirements. Inspections are evidence-driven, not passive. Findings may lead to observations, recalls, or enforcement. Detailed documentation supports FDA decisions, making inspection readiness critical beyond internal review.
Investigates Beyond the Facility
OII conducts investigations beyond facility inspections, covering complaints, product tampering, health fraud, and compliance failures. Activities may occur at manufacturing sites, distribution centers, retail locations, or consumer residences. They gather evidence to support regulatory decisions and coordinate with the Office of Criminal Investigations when potential violations involve illegal activity.
Screens and Controls What Enters the U.S. Market
OII screens FDA-regulated imports at ports, airports, and borders, reviewing foreign products for compliance. Millions of potentially violative items are stopped annually, including drugs and medical devices. For global manufacturers, noncompliance can block market entry, disrupt supply chains, and limit access to the U.S. market.
Operates Continuously and Globally
OII operates globally across more than 90 countries and throughout the United States, overseeing hundreds of thousands of establishments. They conduct tens of thousands of inspections and investigations annually, respond to emergencies, and collaborate with international regulators. FDA oversight is continuous—manufacturers must operate knowing compliance is actively monitored and enforced at all times.

Who Conducts FDA Inspections
FDA inspections are conducted by investigators from the Office of Inspections and Investigations (OII). However, FDA also has a separate criminal enforcement arm—the Office of Criminal Investigations (OCI). OCI special agents are federal law enforcement officers who carry firearms and investigate criminal violations such as fraud, falsification of records, counterfeiting, and illegal distribution.
Their role is distinct from routine inspections. While OII evaluates compliance, OCI investigates potential criminal activity. If OCI is involved, the situation has moved beyond standard inspection or compliance matters.
How FDA Actually Conducts Inspections
FDA inspections follow a structured, evidence-driven approach—not a checklist. Investigators trace data across systems, compare records, and test consistency between what is written and what is actually done. They will move across departments, follow CAPA, review validation, and challenge decisions.
Inspections are dynamic. Questions evolve based on what is found. A single inconsistency can expand the scope quickly. Investigators are not looking for perfection—they are assessing control, traceability, and credibility of your system.
What matters is not how your system is designed, but how it performs under scrutiny. Preparation must reflect how FDA actually inspects—not how you think they inspect.
What to Do When an FDA Investigator Arrives
You do not control when FDA arrives or how long they will stay. An inspection may last a day or extend for weeks or months. The duration is driven by what investigators find as they follow your data, records, and responses.
Their objective is not unclear—they are there to determine whether your operations are in control. Every request, every question, and every document supports that conclusion.
Your role is to demonstrate consistency, organization, and credibility. Follow your inspection protocol, manage communication, and provide accurate, complete information. Do not rush, speculate, or improvise.
You cannot influence how long they stay—but you can influence what they see.
What Are FDA Investigators Actually Looking For?
FDA investigators are not there to “check quality” or review procedures for completeness. Their role is to enforce the law. They evaluate whether your operations comply with applicable regulations and whether your systems produce reliable, controlled, and traceable outcomes.
Procedures alone do not demonstrate compliance. Investigators assess whether your actions match your procedures, whether your data is credible, and whether decisions are supported by objective evidence.
They are looking for evidence of control. Can you trace a decision from start to finish? Does your data support your conclusions? Are deviations investigated properly, or just closed?
Investigators follow records, compare timelines, and look for inconsistencies. If your documentation conflicts, if actions are not justified, or if trends are ignored, that becomes a problem.
This is not about how your system is written. It is about whether your records, actions, and outcomes align under detailed review.
What to Do and Not to Do
Do:
- Verify credentials and expect Form FDA 482 at the start of a domestic inspection.
- Have the most responsible individual or a trained designee meet the investigator.
- Assign a knowledgeable escort to accompany the investigator throughout the inspection.
- Provide requested records promptly, or clearly explain the reason for any delay.
- Ask at the end of each day what was covered and whether deficiencies were noted.
- Correct obvious issues immediately when appropriate, since FDA states prompt corrections made in the investigator’s presence can be viewed positively.
Do not:
- Refuse entry, block access to records FDA is entitled to review, or otherwise interfere with the inspection; FDA’s IOM treats those as refusal issues with potential escalation.
- Photocopy or photograph FDA credentials unless the investigator expressly allows it; the IOM tells investigators not to permit photocopying of credentials.
- Delay just because key staff are busy or records are inconvenient to retrieve.
- Assume the inspection scope is fixed; FDA notes inspections may expand based on what investigators find.
What to Say and Not to Say
Say:
- “Let me verify that and get you the correct record.”
- “Those records are stored off-site; we can retrieve them by tomorrow.”
- “Our designated contact will coordinate this request.”
- “At the end of today, can you tell us what areas you covered and whether you noted any deficiencies?”
These responses match FDA’s own emphasis on open communication, accurate record production, and structured interaction during inspections.
Do not say:
- “I think…”
- “Probably…”
- “We usually…”
- “That’s how we’ve always done it.”
- “We don’t have that.”
- “You can’t take that photo.”
- “Come back later when our expert is here.”
Those answers create risk because FDA inspections are evidence-based, and unsupported statements, speculation, or access limitations can undermine credibility or be treated as interference depending on the circumstance. FDA’s materials make clear that investigators assess what records show, what employees actually do, and whether the inspection can proceed in a reasonable manner.
Best rule:
Answer truthfully, briefly, and only to what was asked. If you do not know, do not guess—say you will verify and return with the correct information. That is usually safer than improvising. This last sentence is a practical inference from FDA’s inspection approach and emphasis on records, consistency, and open communication.
How to Set Up Front Room and Back Room During an FDA Inspection
Large manufacturers do not manage inspections casually. They use a structured front room / back room model to control communication, ensure accuracy, and protect the integrity of responses. This separation allows the company to stay organized while maintaining a consistent, professional interaction with FDA investigators. The front room manages all direct communication with FDA, while the back room supports by gathering records, verifying information, and preparing responses.
This is not about hiding information—it is about controlling flow, ensuring accuracy, and avoiding inconsistent or incomplete responses. Companies that use this model effectively reduce confusion, prevent misstatements, and maintain credibility throughout the inspection.
Manage Requests With Speed and Control
When FDA requests a large set of records, do not wait to assemble everything before responding. Provide what is verified and ready as soon as possible, and clearly communicate that the remaining documents are in progress. This approach demonstrates responsiveness and control without sacrificing accuracy.
In practice, investigators often begin reviewing what is provided immediately. In some cases, initial records may satisfy their questions, reducing the need to review the full set. However, this should never be assumed.
Speed matters—but only with control. Every document must still be accurate, complete, and reviewed before release. The goal is to maintain momentum while ensuring everything provided can withstand scrutiny.
Front Room (FDA-Facing Team)
The front room is where all interaction with the FDA investigator occurs. It typically includes a designated host (often Quality or Regulatory leadership), subject matter experts as needed, and a trained coordinator managing requests. Only controlled, verified information should be presented in this room.
Large companies limit who speaks. One voice leads, others support when called. Every question is listened to carefully, clarified if needed, and logged. No guessing, no off-the-cuff responses. If information is not immediately available, the response is: “We will confirm and follow up.”
The front room sets the tone—professional, organized, and controlled. It demonstrates that your company understands inspection discipline and manages communication deliberately.
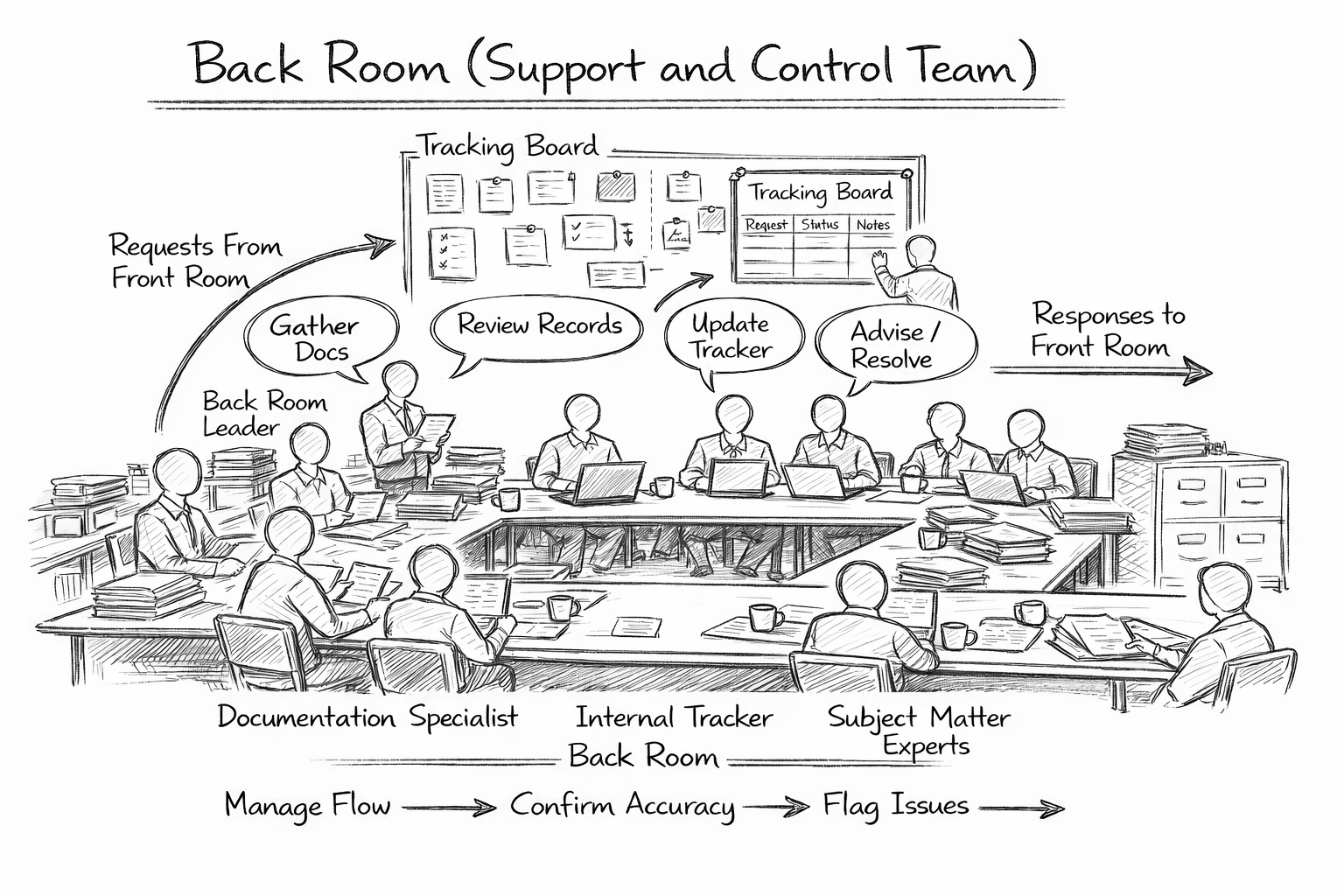
Back Room (Support and Control Team)
The back room operates behind the scenes. This team gathers documents, reviews records, verifies accuracy, and prepares responses before anything is provided to FDA. It includes subject matter experts, document control, and quality personnel who understand the system in detail.
Every request from FDA is tracked, prioritized, and reviewed before release. Documents are checked for completeness, consistency, and potential risk. If gaps are identified, they are escalated internally—not exposed prematurely.
The back room prevents errors. It ensures that what reaches the front room is accurate, aligned, and defensible. This structure allows large organizations to respond quickly while maintaining control over the information being presented.
Minimum Roles During the FDA Inspection
An FDA inspection is not managed by one person—it requires a structured team with clearly defined roles. Each role exists to maintain control, consistency, and speed. Without this structure, communication breaks down and risk increases.
- Executive Presence (Intro & Closing Meeting). It is advisable for the top manager or CEO to attend the opening meeting. This signals seriousness, accountability, and organizational commitment to compliance. It sets the tone immediately.
- Receptionist (First Contact Control). The receptionist controls the first interaction. Minimal conversation only. While notifying the team, they can politely ask the investigator to view a safety video or wait briefly—this creates time to activate the inspection protocol.
- Front Room Leader. Leads all communication with FDA. Controls who speaks, how responses are delivered, and ensures consistency. This is the voice of the company during inspection.
- Back Room Leader. Manages document flow, assigns tasks, verifies accuracy, and ensures responses are complete before reaching the front room. This is the control center.
- Scriber. Documents everything in real time—questions, responses, records provided, and timing. No speaking. Only recording.
- Escort. Stays with the investigator at all times. Controls movement, ensures safety compliance, and observes what the investigator is reviewing.
- Runner (Document Delivery). A dedicated person responsible for moving documents between the back room and the front room. Must be trained, fast, and able to read the situation—timing matters.
- Subject Matter Experts (SMEs). Provide technical answers when called. Must stay within scope, avoid speculation, and respond with controlled, verified information only.

Establish an Internal FDA Inspection Playbook and Train Your SMEs
An FDA inspection playbook must be formalized as a controlled procedure, not an informal practice. It should define roles, communication flow, document control, escalation paths, and how requests are managed from receipt to response. This includes clear expectations for front room and back room operations.
Subject matter experts (SMEs) must be trained on how to respond—not just what they know. Technical knowledge alone is not enough. SMEs should understand how FDA asks questions, how to stay within scope, and how to provide accurate, controlled responses.
Training should include mock inspections, real scenarios, and communication discipline. A prepared SME demonstrates control. An unprepared SME creates risk—even with strong systems.
Prepare Front Desk and Maintain SME Contact Control
The inspection starts at the front desk. The receptionist should not verify credentials or engage in discussion. Their role is minimal and controlled: remain professional, limit interaction, and immediately notify the designated primary and secondary contacts. The only response should be, “Someone will be here shortly.”
At the same time, the receptionist must observe and report key details: how many FDA personnel arrived, the time of arrival, and any statements made. This information should be relayed exactly as observed—no interpretation, no assumptions.
The receptionist should not answer questions or provide information. All communication must be controlled through the inspection lead.
Control begins at the first point of contact—and it starts with discipline.
Conduct Periodic Internal “FDA Inspection” Drills
Internal FDA inspection drills should mirror real conditions—unannounced arrival, real-time questioning, and controlled document flow. The purpose is not to test procedures on paper, but to evaluate how your team performs under pressure. This includes front room coordination, back room support, and SME response discipline.
When conducting drills, the receptionist should clearly state, “This is a drill,” so the team can engage appropriately. When FDA actually arrives, the message changes—“This is not a drill.” That distinction reinforces urgency and triggers the real inspection protocol.
Drills should be performed periodically, with findings documented and corrected. A compliant system must perform under real conditions—not just exist in documentation.
What Goes Wrong During an FDA Inspection
Most issues during FDA inspections are not caused by lack of procedures—they are caused by breakdown in execution. Companies often appear prepared on paper but lose control in real time. Common failures include inconsistent responses between SMEs, delays in retrieving records, and providing unverified or incomplete information.
Communication is another major risk. Speaking too much, guessing, or contradicting documentation can quickly escalate the scope of the inspection. Poor coordination between front room and back room leads to confusion and loss of credibility.
Investigators follow inconsistencies. One weak answer can lead to deeper review, expanding the inspection beyond its original scope.
What goes wrong is rarely technical—it is operational. The inability to demonstrate control, consistency, and discipline under inspection conditions is what creates risk.

FDA Forms 482, 483, and 484 — What Are They?
FDA inspections are formally documented using standard forms. Understanding these forms is critical because they define the inspection process, what FDA observed, and how the inspection is closed. These are not administrative paperwork—they carry regulatory significance and can impact enforcement decisions.
Form FDA 482 Notice of Inspection
Form FDA 482 is issued at the start of a domestic inspection under FDA’s authority (FD&C Act §704). It formally notifies the firm that an inspection is being initiated. Once presented, the inspection has officially begun.
For foreign manufacturers, Form FDA 482 is generally not issued. FDA conducts foreign inspections under different authorities and practices, and the 482 is typically not used in the same way as it is for domestic inspections.
Form FDA 483 Inspectional Observations
Form FDA 483 is issued at the conclusion of an inspection when investigators observe conditions that may violate FDA regulations. It lists specific observations based on what was seen during the inspection.
If no observations are identified, a 483 is not issued. However, absence of a 483 does not mean you are free from future regulatory or legal action. Likewise, receiving a 483 does not mean you “failed” the inspection. It reflects observations that require evaluation and, where appropriate, corrective action.
A 483 is an inspection outcome—not a final determination.
Form FDA 484 Receipt for Samples
Form 484 is issued when FDA collects samples during an inspection. It documents what was taken, including product samples or materials, and serves as an official record of the collection.
Affidavit
An affidavit is a written statement of facts that is confirmed and signed under oath, typically before a notary or authorized official. During an FDA inspection, an investigator may request an affidavit to formally document statements, explanations, or events related to the inspection. This is not routine documentation. An affidavit becomes part of the official record and may be used in regulatory or legal proceedings.
If you are asked to provide or sign an affidavit, treat it seriously. Ensure the content is accurate, complete, and reviewed internally. When appropriate, consult legal counsel before signing. Once executed, it represents a sworn statement that carries legal weight.
A Well-Trained, Silent Scriber Is Essential
A dedicated scribe should document the inspection in real time—accurately and without participating in the discussion. This role is not optional. The scribe captures every request, response, document provided, time stamps, and key statements from the investigator.
The scribe must remain silent, objective, and disciplined. They do not answer questions, interpret responses, or engage with the investigator. Their sole responsibility is to create a clear, factual record of what occurred during the inspection.
These notes become critical after each day’s close and at the end of the inspection. They support internal alignment, help identify potential observations early, and ensure consistency in responses.
If you need structured scribe training—remote or onsite—contact CIRG.
Conduct Annual or Biannual Mock FDA Inspections
Do not wait for FDA to test your system. Schedule annual or biannual mock FDA inspections to evaluate how your organization actually performs under real conditions. These are not routine audits—they replicate FDA behavior, including questioning, record tracing, and cross-functional pressure.
A properly conducted mock inspection will expose gaps in execution, not just documentation. It tests your front room discipline, back room control, and SME response under scrutiny. More importantly, it shows whether your system holds when challenged—not when reviewed internally.
Working with experienced FDA consultants brings the perspective of how inspections are actually conducted. The outcome is not just findings—it is clarity on your true readiness.
If you are serious about inspection readiness, test it before FDA does.